
Contributions
Abstract: S1594
Type: Oral Presentation
Presentation during EHA23: On Sunday, June 17, 2018 from 09:00 - 09:15
Location: Room A10
Background
Mature blood cells of multiple lineages are constantly replenished throughout life. In humans, definitive haematopoiesis in the fetal liver (FL) commences at around 5 weeks of gestation, which remains the main site of haematopoiesis throughout fetal life. Haematopoiesis in the bone marrow (BM) starts around 11-12 weeks of gestation, but does not take over as the primary site of haematopoiesis until just after birth. Currently, we know very little about how haematopoietic stem/progenitor cell (HSPC) subsets change through ontogeny; and whether they do so in a site and stage-specific manner. Single cell RNA sequencing (scRNASeq) techniques now offer unprecedented opportunities to finely dissect the heterogeneity within specific HSPC populations and compare the cellular architecture of different sites of haematopoiesis.
Aims
To characterise the human HSPC compartment from early fetal life to adulthood using scRNAseq.
Methods
Lineage-negative, CD34+ cells from first trimester FL (n=2; gestation week 7-8); matched second trimester FL and fetal BM (FBM) (n=2 each; week 18-19); and healthy adult donor mobilised apheresis HSPC (n=2) were FACS-isolated for scRNAseq using the Chromium 10X platform. Data was processed using CellRanger, Seurat, Partek Flow® packages and in-house pipelines to perform dimensionality reduction, unsupervised clustering (tSNE) and to generate KNN graph differentiation trajectories.
Results
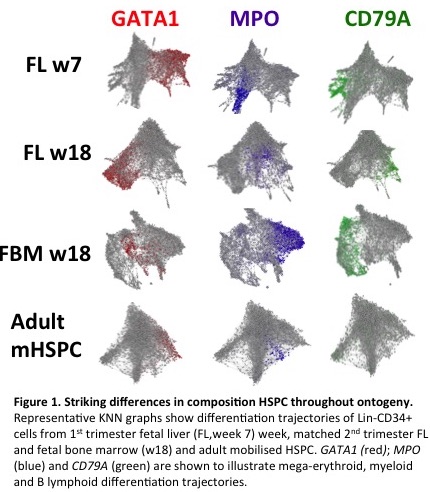
Data was obtained from 5000-9000 cells per sample. After filtering and quality control, data from around 49000 cells from the 8 samples was used for comparative analysis. The mean read count/cell was 47000 and the median gene count/cell was 2500 with 66-70% mapping. The composition of Lin-CD34+ cells was clearly distinct in different stages of ontogeny, with the early FL samples showing a substantially higher proportion of megakaryocyte-erythroid (MkE) progenitors, which decreased during development and in adult life (Fig 1). HSPC composition also varied in a site-specific manner as evidenced by differences seen in matched 2nd trimester FL and FBM samples from the same fetus. Second trimester FBM showed a B lymphoid bias compared to all other tissues including the matched FL samples. Further characterisation of normal and abnormal postnatal BM is currently underway.
Conclusion
We have used scRNAseq to analyse the CD34+ HSPC compartment throughout human ontogeny for the first time. There is clear evidence that this compartment varies in its composition and differentiation potential in a site and developmental stage-specific manner, as it may be dependant on the physiological processes/ demands of that particular developmental stage; and/ or in response to specific microenvironmental cues. Studying haematopoiesis throughout the human lifespan may be important not only to understand normal developmental processes, but also to understand the pathogenesis of postnatal haematological diseases that may have their origins in fetal life.
Session topic: 24. Hematopoiesis, stem cells and microenvironment
Keyword(s): Hematopoiesis, Ontogeny, Stem and progenitor cell
Abstract: S1594
Type: Oral Presentation
Presentation during EHA23: On Sunday, June 17, 2018 from 09:00 - 09:15
Location: Room A10
Background
Mature blood cells of multiple lineages are constantly replenished throughout life. In humans, definitive haematopoiesis in the fetal liver (FL) commences at around 5 weeks of gestation, which remains the main site of haematopoiesis throughout fetal life. Haematopoiesis in the bone marrow (BM) starts around 11-12 weeks of gestation, but does not take over as the primary site of haematopoiesis until just after birth. Currently, we know very little about how haematopoietic stem/progenitor cell (HSPC) subsets change through ontogeny; and whether they do so in a site and stage-specific manner. Single cell RNA sequencing (scRNASeq) techniques now offer unprecedented opportunities to finely dissect the heterogeneity within specific HSPC populations and compare the cellular architecture of different sites of haematopoiesis.
Aims
To characterise the human HSPC compartment from early fetal life to adulthood using scRNAseq.
Methods
Lineage-negative, CD34+ cells from first trimester FL (n=2; gestation week 7-8); matched second trimester FL and fetal BM (FBM) (n=2 each; week 18-19); and healthy adult donor mobilised apheresis HSPC (n=2) were FACS-isolated for scRNAseq using the Chromium 10X platform. Data was processed using CellRanger, Seurat, Partek Flow® packages and in-house pipelines to perform dimensionality reduction, unsupervised clustering (tSNE) and to generate KNN graph differentiation trajectories.
Results
Data was obtained from 5000-9000 cells per sample. After filtering and quality control, data from around 49000 cells from the 8 samples was used for comparative analysis. The mean read count/cell was 47000 and the median gene count/cell was 2500 with 66-70% mapping. The composition of Lin-CD34+ cells was clearly distinct in different stages of ontogeny, with the early FL samples showing a substantially higher proportion of megakaryocyte-erythroid (MkE) progenitors, which decreased during development and in adult life (Fig 1). HSPC composition also varied in a site-specific manner as evidenced by differences seen in matched 2nd trimester FL and FBM samples from the same fetus. Second trimester FBM showed a B lymphoid bias compared to all other tissues including the matched FL samples. Further characterisation of normal and abnormal postnatal BM is currently underway.

Conclusion
We have used scRNAseq to analyse the CD34+ HSPC compartment throughout human ontogeny for the first time. There is clear evidence that this compartment varies in its composition and differentiation potential in a site and developmental stage-specific manner, as it may be dependant on the physiological processes/ demands of that particular developmental stage; and/ or in response to specific microenvironmental cues. Studying haematopoiesis throughout the human lifespan may be important not only to understand normal developmental processes, but also to understand the pathogenesis of postnatal haematological diseases that may have their origins in fetal life.
Session topic: 24. Hematopoiesis, stem cells and microenvironment
Keyword(s): Hematopoiesis, Ontogeny, Stem and progenitor cell


